  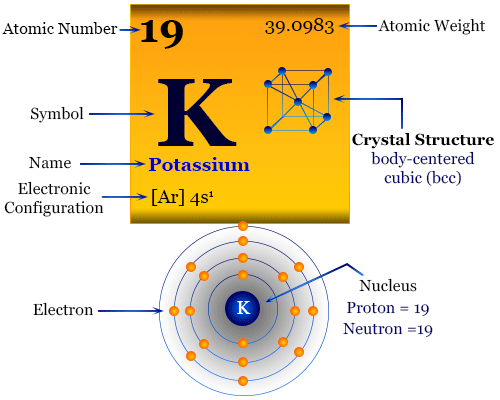
This chemical is most commonly found in artificial fertilizers. The most significant potassium component is potassium chloride (KCl).Potassium may also produce potassium halides through fast reactions with all halogens (F2, Cl2, I2) (KF, KCl, KBr).When potassium is burnt in the air, it can produce the orange potassium superoxide KO 2.Potassium may also generate oxide layers when it reacts with air. The extreme heat produced by this reaction, along with the H 2 as a result, has the potential to be explosive.It may aggressively react with water to generate hydrogen (H 2) gas. Potassium, like other alkali metals, is highly reactive.Potassium salts are formed when the cationic form of potassium mixes with various anions. Like other alkali metals, it contains a single valence electron that is easily removed, resulting in the formation of a cation.Potassium’s electron configuration is 4s1. It has the atomic number 19 and is an alkali metal. Potassium (K) is found in Group 1 and Period 4 of the periodic table.Caustic potash, another key potassium source, is mined mainly in Germany, New Mexico, California, and Utah. These minerals are frequently discovered in ancient lake and sea bed sediments.MgCl 2♶H 2O), langbeinite (K 2Mg 2(SO 4) 3), and polyhalite (K 2Ca 2Mg(SO 4) 4♲H 2O)).Potassium is found in the minerals sylvite (KCl), carnallite (KCl.Davy used the same process to separate sodium a few months after finding potassium. Sir Humphry Davy obtained metallic potassium in 1807 by electrolysis of molten caustic potash (KOH). 
Although potassium is the eighth most plentiful element on the planet, accounting for around 2.1% of the earth’s crust, it is a highly reactive element that is never found free.Humphry Davy, a British chemist, isolated it for the first time in 1807 from potash electrolysis (KOH).The letter K is derived from the Latin word “ kalium,” which derives from the Arabic phrase “ qali,” which means alkali.Because it is found in caustic potash, the term originates from the English “potash” or “pot ashes” (KOH).See the fact file below for more information on Potassium, or you can download our 29-page Potassium worksheet pack to utilize within the classroom or home environment. Potassium also aids muscular contraction and maintains appropriate blood pressure. Its counterpart, sodium, maintains normal fluid levels outside of cells. Potassium can be found naturally in many foods and as a supplement. It is an alkali metal and is solid at ambient temperature. Potassium has the chemical symbol K and the atomic number 19. Are there any risks associated with too much potassium consumption?.What are the benefits of having more potassium in my diet?.Frequently Asked Questions About Potassium.Download the Potassium Facts & Worksheets. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
